Lasers and Clean Energy
Can advanced laser diagnostics help to develop metal fuels as a replacement to fossil fuels? (LLC, Sweden)
One of the most important things for mankind is a safe, reliable and sustainable energy supply. At the moment, ≳ 80 % of the worldwide energy utilisation comes from combustion, where fossil fuels, e.g. coal, gasoline, diesel, are in the majority. Clearly, due to the limited resources and environmental impact, mainly from the large emission of carbon-dioxide, these fossil fuels have to be phased out in the near future.
An alternative fuel, which has seen little consideration thus far, is the use of metal powders, e.g. iron, aluminium or silicon. These fuels have the great advantages that the energy density is higher than those of fossil fuels, the combustion takes place without any emis-sion of carbon-dioxide, polycyclic aromatic hydrocarbons (PAH) or soot, and furthermore the fuel is transportable and relatively safe to store. Clearly, as a new fuel there are many aspects that are unknown and which have to be investigated in detail in the laboratory before this fuel can be used by society to replace fossil fuels. As part of this necessary development, the use of advanced laser diagnostic techniques can be instrumental. These techniques can provide non-intrusive measurements with high spatial and temporal resolution. The parameters that are of primary importance are species concentration, temperature, flow velocity and particle characteristics.

Burning iron particles
The Division of Combustion Physics within Lund Laser Centre (LLC) has for many years been developing and ap-plying different laser diagnostic techniques for character-ising various combustion phenomena. Thanks to recent financial support, it has also been possible to develop and apply laser techniques for further development and understanding of metal combustion. When using laser di-agnostics in a particle environment, special attention has to be paid to the strong Mie scattering as well as multiple scattering which may spectrally interfere. However, similar challenges are seen when working in conventional fuels, e.g. in spray combustion. A technique based on structured illumination can be used here: structured laser illumination planar imaging, whereby the multiple scattering can be suppressed. Furthermore, a new development of conventional Raman scattering based on periodic shadowing, can be applied. When using this technique, a development to suppress background based on lock-in of polarisation rotation as well as time gating can also be utilised. Another approach is to use various coherent techniques, e.g. coherent anti-Stokes Raman, degenerate four-wave mixing or laser-induced grating spectroscopy. Here, the signal is generated as a new laser beam and all particle scattering can be simply suppressed using apertures.
So to summarise, by using appropriate laser diagnos-tics and in close collaboration with modelling expertise in computational fluid dynamics and chemical kinetics and with industry, the aim and hope is to develop these new and fascinating fuels to the benefit of our society and for our environment.
Marcus Aldén (LLC)
Enhanced hydrogen production through electrolysis by novel, laser-induced nanostructured nickel electrodes (ULF-FORTH, Greece)
Researchers from the Ultraviolet Laser Facility at FORTH (ULF-FORTH) in collaboration with the Laboratory of Matter Structure and Laser Physics at the Technical University of Crete (LMSLP-TUC) have initiated experimental research using ultrashort laser pulses to fabricate 2D and 3D nano-structures for applications in sustainable, carbon-free and eco-friendly energy production and storage. An important milestone towards this goal is to produce nanostructured electrodes for use in hydrogen production through electrolysis or for energy storage in batteries.
Nickel is among the top materials to be used as the template to produce the laser-nanostructured electrodes for micro-batteries. In this work, laser structured nickel electrodes are produced and tested for the production of hydrogen gas in alkaline electrolysis. The nickel surface was laser-scanned using optimal conditions (fluence, pulse number per spot) for production of laser-induced periodic surface structures (LIPSS, or “ripples”). LIPSS are known to be the result of the interaction of the incident laser field with a surface wave, created by the laser itself (the surface plasmon). The spacing of the lines produced during the scanning of the metallic surface with the laser beam was chosen to ensure that the metallic surface was densely covered by the ripple nanostructures, i.e. slightly overlapping scan lines. A scanning electron micrograph (SEM) of the laser-scanned metallic surface is shown in Figure 1. Almost homogeneous ripples are shown to cover an extended metallic surface. The ripples form a periodic pattern with a period that is dictated by the parameters of the incident laser field (wavelength, intensity). By performing a Fourier transform on the SEM image, shown at the inset of Figure 1, the period of the nanostructured ripples was found to be 460 nm with a relatively narrow distribution.
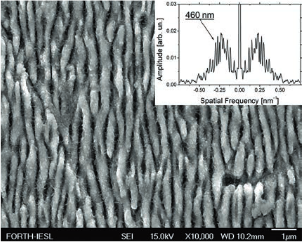
Figure 1: SEM image of the laser-nanostructured nickel surface. Inset shows the Fourier transform of the SEM image
A custom-made electrolysis cell was set up in order to evaluate the performance of the nanostructured electrodes in the production of hydrogen, during the hydro-gen evolution reaction (HER). The electrolyte was 1 Mole potassium hydroxide solution, the anode was untreated nickel and the potential was kept steady at 3 V. It was found that the densely nanostructured electrode produces 3.7 times more hydrogen than an untreated (flat) one (Figure 2). This increase is due to the creation of periodic nano-structures and additional nanoparticles on the electrode surface created by the laser interaction. These increase the electrochemical active surface area, which in turn leads to an increase at the production rate of hydrogen gas.This approach has demonstrated that laser structured nickel electrodes do produce larger quantities of hydrogen gas, and that they can be used not only as hydrogen storage electrodes but also as cathode electrodes in alkaline electrolysis HER.
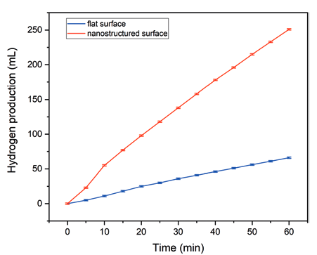
Figure 2: Hydrogen gas production comparison between an untreated nickel electrode and a laser-nanostructured electrode, for one hour of electrolysis.
This approach may help the long or short scale timeline of hydrogen production, because of its low cost, short time of manufacturing and the large quantities of produced hydrogen. Further work is underway in order to optimise the electrode characteristics for optimally efficient hydrogen production or for use in energy storage in batteries.
Panagiotis Loukakos, Maria Farsari, Michail Tsanakas, Nicandra Papakosta, Argyro Klini (FORTH), Ioannis Poimenidis, Stavros Moustaizis (TUC)
Ultrafast optical spectroscopy sheds light on the primary steps of solar fuel production (CUSBO, Italy)
Within the European Green Deal, a key challenge is to decarbonise the energy sector and provide renewable, clean energy. Sunlight is a potential long-term solution: ≈ 0.05 % of incident solar energy would meet current global needs. However, harvesting this energy must be both efficient and cost-effective.
For the two approaches to solar harvesting (photo-voltaic and photocatalytic), a key aspect is the development of novel materials with higher efficiencies or simpler production processes. For photocatalysis, the focus is on nanostructured materials, in which light absorption is followed by charge separation across an interface. This is then used to drive a redox reaction, e.g. reducing H20 into hydrogen. Ultrafast optical spectroscopy plays a key role in these research efforts, as it illuminates both the mechanics of photoexcitation in molecules/materials and the real-time light-to-energy conversion process.
Semiconductor-metal hybrid nanoparticles (HNPs), in which a semiconductor nanostructure is coupled to a noble-metal co-catalyst, offer a highly controllable platform for light-induced charge separation and photocatalysis [1]. The many degrees of freedom (shape, size and composi-tion of both semiconducting and metal components) al-low tunable electronic and optical properties, but also require careful optimisation.
Prof. Uri Banin’s group (Hebrew University of Jerusa-lem), together with the team at the Centre for Ultrafast Sci-ence and Biomedical Optics (CUSBO), Politecnico di Milano, used ultrafast spectroscopic tools to decipher the complex charge transfer pathways in HNPs consisting of semiconductor nanorods with gold tips (see figure below). First, the CdSe-Au HNP model system was used to study the effect of the tip size on photocatalytic function. Combining transient absorption, hydrogen evolution kinetics, and theoretical modelling revealed a non-monotonic behaviour with the size of the gold tip [2]. Considering the size-dependent interplay of the metal domain charging, the relative band-alignments, and the resulting kinetics, an optimal tip size was found. Using nonlinear excitation, in which multiple excitons are generated in the semiconducting component, revealed a competition between Auger recombination and charge separation: the dominant process switched from Auger recombination to charge separation with increasing size of the metal domain, allowing for effective multiexciton dissociation and harvesting in large-metal-domain HNPs, and an improvement of their photocatalytic activity [3].
Very recently, CdSe-Au HNPs was studied because it manifests strong hybridisation of the excitonic and plasmonic states. In these systems, charge separation can occur following photoexcitation of either the semiconductor or the localised surface plasmon resonance (LSPR) of the metal. In particular, a novel mechanism of plasmon induced charge transfer (PICT) was discovered, whereby the LSPR exci-tation is directly accompanied by a rapid charge separation, creating an electron in the semiconductor and a hole in the metal. By applying spectroscopy with >10-fs resolution, the complete pathway of electron transfer was resolved for both semiconductor and LSPR excitation. The LSPR experiences an ultrafast (< 30 fs) electron transfer to CdSe via PICT, followed by back transfer from the semicon-ductor to the metal within ≈ 200 fs (see figure).
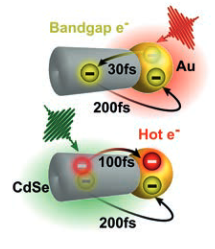
Ultrafast optical spectroscopy is used to visualise in real time the forward and backward electron transfer processes in metal-semiconductor hybrid nanoparticles, which occur on the 100-fs timescale.
These studies show the power of ultrafast optical spec-troscopy to identify the complex nanoscale photophysical processes involved in solar energy conversion in HNPs and to measure the corresponding timescales. These results establish the design parameters for HNPs in a photocatalytic material.
Giulio Cerullo, Francesco Scotognella,Sandro De Silvestri (CUSBO) and Uri Banin (Hebrew University Jerusalem)
[1] P. Kamat, J. Phys. Chem. Lett. 3: 663–672, 2012
[2] Y. Ben-Shahar et al., Nat. Commun. 7: 1–7, 2016
[3] Y. Ben-Shahar et al., Nano Lett. 18: 5211-5216, 2018
Using time-resolved spectroscopy to identify the excited states leading towards hydrogen generation (CLF, UK)
New approaches to the generation of sustainable energy sources are high on political agendas. This is for a range of reasons, including climate change, depleting fossil fuels, and a predicated increase in energy requirements. Currently, the main source of energy is fossil fuels, which when burned release pollutants and most importantly green-house gases. An alternative approach for energy generation is the use of sunlight. This can be harnessed by using photosensitisers to split water into hydrogen and oxygen.An aim of the research of Dr Mary Pryce (Dublin City University, Ireland) in collaboration with Dr Elizabeth Gibson (Newcastle University) is to devise new, robust, cheap, and abundant photocatalytic materials that are capable of splitting water into hydrogen and oxygen. In this way, solar energy can be effectively stored as a solar fuel (H2). Time-re-solved techniques, such as time-resolved absorption spectroscopy (TA) and time-resolved infrared spectroscopies (TRIR), are essential tools in characterising the excited states leading to hydrogen generation from water. For example, in a recent Laserlab-Europe access project (EU access grant 16140001 and CLF002565) with the UK Central Laser Facility (CLF), they used both TRIR and TA to identify the excited states generated in BODIPY copolymers with conjugated comonomers [1][2]. They used these polymers to generate hydrogen from water in the presence of a cobalt catalyst.
The time-resolved techniques provided important insights into the reactivities of reactive intermediates in the hydrogen generation process. Armed with this data, it is possible to design and develop superior photocatalytic materials, to enhance the efficiency of hydrogen generation.
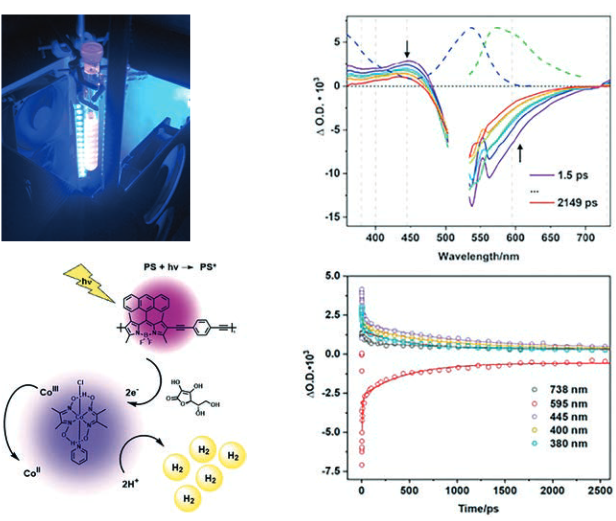
Using time-resolved spectroscopy to identify the excited states leading towards hydrogen generation. (c) Mary Price
In addition to this water-splitting research, UK-based research groups have used the facilities at the CLF to apply time-resolved techniques to track the reaction pathways in Earth-abundant organometallic [3] and organic [4] homogeneous catalysts. For this both TRIR and pump-IR probe-probe-probe (TRMPS) vibrational spectroscopy have proven invaluable in tracking the reaction pathways of model systems all the way from picosecond to millisecond timescales. The knowledge this provides of the steps involved in the catalytic cycles will allow rational approaches to the optimisation of catalysts. The new mechanistic understanding gained helps in the aim of developing Earth-abundant metals as competitive replacements for precious metal catalysts, as well as driving towards cleaner and cheaper means of forming industrial chemical precursors and complex molecular architectures.
Mary Pryce (DCU) and Mike Towrie (CLF)
[1] A. A. Cullen et al., Front. Chem. 8: 957, 2020
[2] N. Põldme et al., Chem. Sci. 10: 99-112, 2019
[3] L. A. Hammarback et al., Nat. Catal. 1: 830-840, 2018
[4] A. Bhattacherjee et al., Nat. Commun. 10: 5152, 2019
Pump-pump-probe spectroscopy sheds light on key reactions for artificial photosynthesis (ISMO, France)
Artificial photosynthesis aims at producing energyrich chemical compounds, such as H2 or methane, from H2O and CO2 using sunlight. Several steps have to be reproduced: sunlight must be captured, and electronic excitation must be converted into electronic energy stored in the catalyst using both charge separation and chemical reactions. The main reactions that need to be mimicked are gathered into the oxygen evolution reaction (OER), the hydrogen evolution reaction (HER) and the CO2 reduction reaction (CO2RR).
These photocatalytic reactions, which produce solar fuels, require coupling of multiple photo-induced single-electron transfers (to accumulate charges on the catalytic centre) with multielectron catalysis. Understanding of these lightdriven charge accumulations in photosystems is thus of pivotal importance to guide optimisation of the photosystems. It has been identified as one of the bottlenecks to progress on this solar energy storage technology. A new experimental set-up at Institut des Sciences Moléculaires d’Orsay (ISMO) performs pump-pump-probe transient absorption spectroscopy to access the electronic excited states and their relaxation dynamics from the nanosecond to the millisecond range of timescale, in particular those of the charge accumulation and recombination in multicomponent molecular systems.
This set-up recently demonstrated light-induced two-electron accumulation on a model multicomponent system made of the [Ru(bpy)3]2+ molecule as the light absorber, ascorbate as the reversible electron donor and naphthalene diimide as the two-electron acceptor. Accu-mulative charge separation was observed in both single-pulse pump-probe and double-pulse pump-pump-probe experiments. The doubly reduced state was produced with up to 4.2 % yield and revealed a lifetime of about 100 microseconds. As far as it can be ascertained, this is the first demonstration of charge accumulation with only a single-pulse excitation in a multicomponent system. Time resolved resonance Raman spectroscopy is also being de-veloped to complement the transient absorption experiments performed in the visible range of wavelengths. This type of investigation is key to mapping the path to artificial photosynthesis.
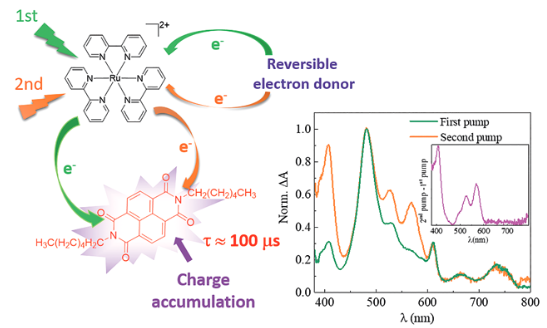
Schematic diagram of the photochemical process, along with a graph of the transient absorption spectrum of the pump-pump-probe pulse used to trigger it.
Performing such experiments in the presence of the substrate, CO2 for instance, it is now possible to investigate the fundamental processes at work in the photocatalytic cycle of specific catalysts for laboratory-scale production of solar-derived fuels. It is noteworthy that one major challenge concerns the development of noble-metal-free, efficient, selective, and recyclable homogeneous or hetero-geneous catalysts. Within the framework of collaborating investigation teams, synthesising photocatalysts in respect to this challenge, new families of hybrid organic-inorganic molecular photosystems are investigated for CO2 reduc-tion (CO2RR) and HER reactions toward solar fuels.
Thomas Pino and Minh-Huong Ha-Thi (ISMO)
T. Tran et al., J.Phys.Chem. C 123: 28651, 2019
How to improve the efficiency and stability of organic dyes for luminescent solar concentrators (LENS, Italy)
Due to their high efficiency (15 – 20 %) and light-harvesting capability, silicon-based solar cells are widespread devices and presently contribute more than 2 % to global electricity generation. In the search for new technologies to boost the output of PV (photovoltaic) cells, the idea of increasing the light intensity by concentration is a potential solution to reduce the required photovoltaic material. Luminescent solar concentrators (LSCs) are a technology in which part of the spectrum of incident sunlight is down-converted by an efficient fluorophore dispersed in a transparent panel and sent to the surface of a PV cell. Due to the difference in refractive index between the panel material and air, most of the light emitted by the fluorophore is concentrated at the edges of the panel, where PV cells can be positioned. Since this concentrates the incident light by a factor of 5-10, without the need for expensive tracking, smaller silicon (or other) solar cells can be used. As the cost per square centimetre of the transparent plastic is lower than that of the solar cell, the cost per watt is lower than the cost of a planar silicon solar cell. Also, LSCs are of special interest for building integrated PV applications (for example in windows and roofs).

A piece of LSC material, showing increased brightness at the hand.
LSCs still have some drawbacks limiting their applications, particularly durability. Big efforts have been invested in developing new fluorophores and in particular, in designing suitable molecules with a wide absorption range (between 450 and 700 nm), high fluorescence quantum yield, large Stokes shift, good solubility in the host polymeric matrix and long optical and thermal stability. Various materials have been proposed and tested as fluorophores: quantum dots, inorganic and organometallic molecules, and small organic molecules such as perylene diimides, BODIPY oligomers, benzothiadiazoles and diketopyrrolopyrrole dyes.
Within this framework, the Ultrafast Spectroscopy group of the Photonic Materials area at LENS, the European Laboratory for Non-Linear Spectroscopy, started new collaborations with industrial laboratories and the Italian National Research Council (CNR), making use of standard spectrophotometric and laser techniques in characterising new organic molecules, aiming to identify the principal parameters making them usable in long term stable LSCs. Of course, materials that have facile and scalable synthetic routes, good processability, and high reproducibility are preferred.Research efforts have been focussed on organic dyes with improved stability and efficiency in terms of fluorescence quantum yields and possessing a large Stokes shift. A combined approach of quantum mechanical calculations and photophysical characterisation of dye molecules has re-sulted in good strategies to achieve efficient fluorophores.
An analysis of the in-vestigated organic fluo-rophores shows that a high radiative rate con-stant, that is, a high photoluminescence quantum yield and a short fluorescence lifetime, is one of the key points. In addition, isomerisation has been shown to worsen the photophysical characteristics of LSCs. Many fluorophores showing good characteristics in terms of efficiency and stability work by charge-transfer transitions. Rigidity of the molecular scaffold or of the surrounding polymer chains can help in increasing stability and making a more robust device. Pump-probe spectra with sub-picosecond pulses suggest that preventing large excited-state conformational relaxations, as happens in the polymeric matrix, results in increased fluorescence quantum yields.
Many of the investigated organic compounds belong to the donor-acceptor and donor-acceptor-donor type of molecules. They are synthetised to possess an intense emission spectrum between 450 – 600 nm, overlapping the light-absorption spectrum of Si-based photovoltaic cells. They down-convert the blue part of the solar spectrum where the efficiency of Si-cells is usually low. This causes the panel to be yellow-red coloured. The Stokes shifts are large (70 – 100 nm) to minimise the self-absorption effects. Recent results confirm the possibility of achieving efficiencies of more than 5 %.
Paolo Foggi (LENS), Mariangela DiDonato (LENS and CNR-ICCOM), Barbara Patrizi (LENS and CNR- INO)
Using simulation, experiments and spectroscopy to improve the design of photocatalysts (IPHT, Germany)
Green plants and many algae obtain their energy from the conversion and storage of sunlight. In natural photosynthesis, light harvesting molecules such as chlorophylls absorb solar energy and convert it into one of nature’s currencies of chemical energy, i.e. adenosine triphosphate (ATP). The plant uses this chemical energy and carbon dioxide (CO2) to produce energy-rich molecules such as sugars, which can be stored over a long time.
Imitating what seems to work effortlessly in nature is still a great challenge for science. Based on the model of natural photosynthesis, the 2018 established Transre-gional Collaborative Research Centre (TRR 234) CataLight (Light-driven Molecular Catalysts in Hierarchically Structured Materials – Synthesis and Mechanistic Studies) researches novel soft-matter systems to convert solar energy into chemical energy carriers such as hydrogen – a renewable and clean fuel. To achieve this, CataLight embeds molecular photocatalysts, the mode of action of which can be tuned by molecular design, into soft-matter photocatalytic systems for the production of hydrogen and oxygen from water, but also for other industrially relevant photooxida-tion and photoreduction reactions [1].
The approach CataLight pursues is to integrate molecular photocatalysts into stimuli-responsive soft matter matrices and to understand the molecule-matrix interactions. The heterogeneous matrices offer vast opportunities to synergistically control charge transfer processes, photo-chemical reaction pathways and degradation resistance. However, the underlying mechanisms, which control component interactions, photochemical and -physical properties and overall catalytic activity are not fully understood yet. In order to make artificial photosynthesis a reality, these basic processes and mechanisms must first be elucidated.
Lasers are versatile and powerful tools to shine light on the complex light‐activated multistep reaction mechanisms that take place in a molecular catalyst for artificial photosynthesis. For example, in ultrafast time-resolved spectroscopy, pulsed lasers allow to picture processes that otherwise could not be observed because they take place on the pico- to femtosecond timescale and involve highly transient reactive intermediates. Combined with other laser-based techniques such as resonance‐Raman spectros-copy, and computational studies, it is possible to deduce important structure-function-activity relationships that hence allow for the design of more efficient catalysts for artificial photosynthesis [2][3].
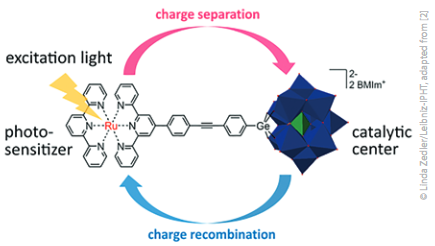
The kinetics of light-induced charge separation and recombination in a photocatalyst determine its efficiency for artificial photosynthesis.
Such a combined experimental, theoretical and spec-troscopic approach was recently used to identify and spectroscopically characterise those highly reactive intermediates and their excited‐state dynamics. Spectroelectrochemical methods (UV/Vis absorption and resonance‐Raman spectroscopy), quantum‐chemical simulations and time‐resolved transient absorption (TA) spectroscopy elucidated the photoinduced electron transfer in the catalytically competent intermediate of a dinuclear transition metal-based photocatalyst for hydrogenation of nicotina-mide (NAD‐analogue) and proton reduction. The in-situ photophysical studies on femto‐to‐nanosecond timescales revealed that electronic transitions shift electron density from the activated catalytic centre to a bridging ligand, which significantly reduces the catalytic activity upon visible‐light irradiation. Based on these results, the photocat-alysts can be synthetically redesigned to be more efficient – a spectroscopically guided approach to make artificial photosynthesis become reality.
Benjamin Dietzek and Anja Schulz (IPHT)
[1] I. Krivtsov et al., Angew. Chem. Int. Ed. 59: 487, 2020
[2] L. Hammarström, Acc. Chem. Res. 48: 840, 2015
[3] F. A. Black et al., Chem. Sci. 9: 5578, 2018

